Scientifically reviewed by
Dr. Ky H. Le, MD

The information presented in this article is for educational and research purposes only, intended for laboratory professionals, researchers and collaborators. This content does not constitute medical or clinical advice.
Follistatin is a well-studied regulating glycoprotein in muscle biology and metabolic science. It inhibits TGF-β binding and performs an essential function in various physiological processes, including muscle regeneration, growth, and glucose regulation.
Its scientific uses are diverse, including skeletal muscle development, adipose tissue modulation, bone growth, cardiovascular function, and cellular signaling pathways.
Key Research Insights
- Follistatin is a binding protein that neutralizes TGF-β superfamily ligands like activin, myostatin, and BMPs.
- According to various studies, follistatin stimulates myoblast fusion, muscle fiber development, and regeneration by activating the PI3K/Akt/mTOR pathway.
- The protein affects adipocyte development, glucose homeostasis, and pancreatic beta cell function in metabolic studies.
- Follistatin inhibits BMP and promotes the differentiation of mesenchymal stem cells.
What is Follistatin Protein?
Follistatin is a secreted glycoprotein that binds to members of the TGF-β superfamily. The protein is primarily found in two isoforms: FST288 and FST315, which originate from alternative splicing.
These isoforms exhibit significant differences in biological behavior and tissue distribution patterns. FST288 has a high affinity for cell-surface heparan sulfate proteoglycans and remains tissue-bound, whereas FST315 has a lesser affinity but circulates more freely in the system[1].
Follistatin regulates biological activity by binding and neutralizing TGF-β family ligands. These ligands include activin, myostatin, and bone morphogenetic proteins (BMPs), which prohibit them from binding to their respective cellular receptors.
Related Product: Buy Follistatin for laboratory research use. Our Follistatin (FLGR242) is a novel fragmented, modified version of Follistatin-344 (FST-344) that does not bind to the protein activin and instead contains a patented albumin binder.
Molecular Mechanisms of Action
Structural studies demonstrate that follistatin envelops activin dimers, with its FSD2 domain obstructing the type II receptor–binding region. This interaction generates stable complexes, each consisting of two follistatin molecules bound to a single activin dimer[2].
Follistatin also exhibits varying ligand affinities. It binds activin A more strongly than bone morphogenetic protein-2 (BMP-2) and is capable of directly associating with several BMPs with notably high affinity.
Myostatin Antagonism
Follistatin inhibits myostatin, a negative regulator of muscle growth, through direct structural interactions. Crystal structure analysis shows that follistatin’s prehelix region and binding domains make extensive contacts with myostatin[3].
Studies further indicate that many of the follistatin residues essential for myostatin interaction overlap with those used to bind activin. The protein can bind circulating myostatin in serum, where the majority of myostatin is found bound to its propeptide or to follistatin-family proteins[4].
Skeletal Muscle Research Applications
Research shows that nitric oxide (NO) and cyclic GMP (cGMP) induce follistatin expression, and this secreted protein mediates their actions in myogenesis. Deregulation of cGMP signaling leads to fusion-induced hypertrophy of satellite-derived myotubes[5].
Studies also reveal that mammalian target of rapamycin (mTOR) regulates follistatin expression during skeletal myogenesis. mTOR induces MyoD-dependent follistatin expression that leads to myocyte fusion[6].
Muscle Fiber Growth Studies
Animal studies reveal that systemic administration of follistatin288 increases lean body mass and muscle mass. Follistatin-overexpressing transgenic animals show substantially greater myofiber regeneration compared to controls[7].
The protein appears to operate through multiple mechanisms including myostatin blockade and blood vessel formation. Research shows follistatin promotes myoblast proliferation by activating the PI3K/Akt/mTOR signaling pathway[8].
Muscle Regeneration Pathways
Following muscle injury, follistatin contributes to healing through interactions with muscle regeneration, angiogenesis, and fibrosis pathways. Animals overexpressing follistatin undergo substantially greater myofiber regeneration and show reduced fibrosis formation after skeletal muscle injury[7].
The protein’s mode of action involves blocking myostatin while at the same time promoting blood vessel formation in damaged tissue. This dual mechanism makes it particularly relevant for muscle repair research.
Metabolic Research Applications
Adipose Tissue and Thermogenesis
Follistatin promotes adipocyte differentiation and influences the browning of adipose tissue. The protein is induced during adipogenic differentiation of brown preadipocytes and mouse embryonic fibroblasts, as well as in cold-induced brown adipose tissue[9].
Studies show follistatin improves thermogenic gene expression in differentiated brown adipocytes. The induction of brown adipocyte proteins including uncoupling protein 1 (UCP1) is attenuated in follistatin knockout cells but can be rescued by recombinant follistatin treatment[10].
Glucose Regulation Studies
Research indicates follistatin and its homologous protein follistatin-like 3 (FSTL3) participate in glucose regulation. Animals lacking FSTL3 develop increased pancreatic islet number and size, beta cell hyperplasia, decreased visceral fat mass, improved glucose tolerance, and improved insulin sensitivity[11].
Follistatin overexpression in muscle increases insulin action, as shown by improved basal and insulin-stimulated glucose uptake[12]. These findings suggest follistatin plays a regulatory role in glucose homeostasis at the cellular level.
Pancreatic Function Research
Follistatin influences pancreatic islet function through its antagonism of activin and related ligands. Neutralization of FSTL3 improves both insulin secretion and glucose responsiveness in non-functional mouse and human islets[13].
Animals with altered follistatin or FSTL3 expression show changes in serum insulin levels and pancreatic insulin content. This positions follistatin as a key regulator in pancreatic cell research.
Bone Development and Repair Research
BMP Modulation Mechanisms
Follistatin functions as an antagonist of bone morphogenetic proteins, which are primary regulators of bone formation. The protein can directly bind to BMP-receptor complexes, showing that follistatin is noncompetitive with the BMP receptor[14].
Among BMP antagonists expressed during bone development, follistatin shows regional-specific expression patterns that correlate with the regulation of mesenchymal progenitor cell proliferation and differentiation[13].
Osteogenesis Studies
Follistatin promotes multiple processes needed for bone repair, including cell recruitment, osteogenesis, and vascularization. Both FST288 and FST315 variants influence mesenchymal stem cell behavior and bone formation[13].
The research shows that follistatin improves mesenchymal stem cell migration and osteogenic differentiation in experimental models. The protein’s ability to bind activin A, which inhibits bone formation, contributes to its osteogenic effects.
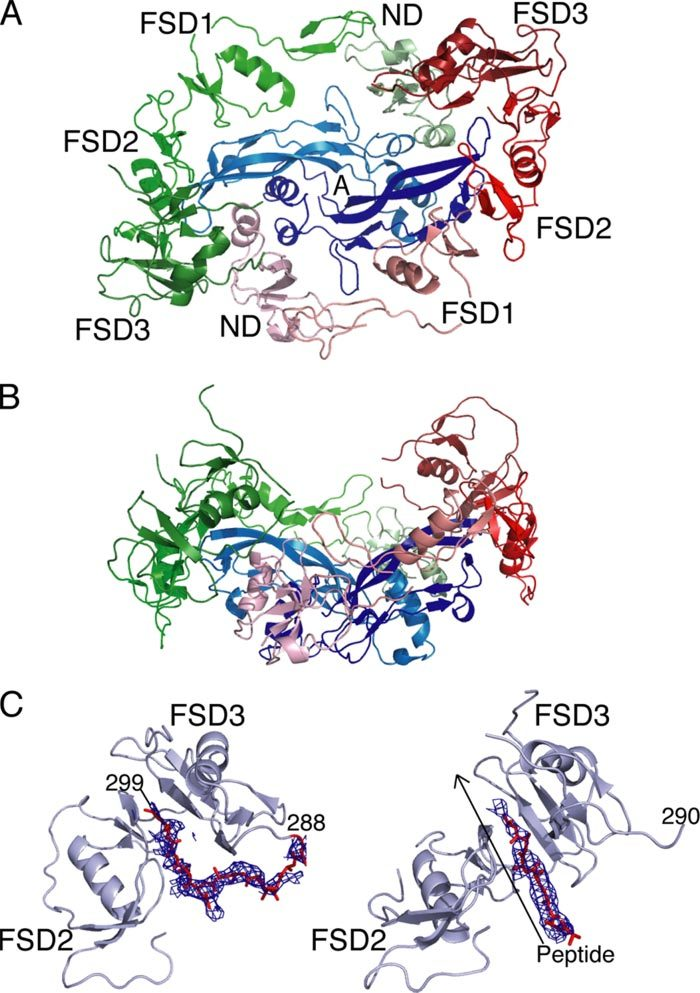
Structure of the activin-follistatin 315 complex (source publication)
Cardiovascular Research Applications
Endothelial Cell Function
Follistatin-like 1 (FSTL1) functions as a secreted muscle protein that promotes endothelial cell function and revascularization in ischemic tissue. The protein activates Akt-endothelial nitric oxide synthase (eNOS) signaling pathways[15].
FSTL1 released from skeletal muscle increases circulating levels and promotes endothelial cell migration and differentiation. Dynamic resistance exercise increases skeletal muscle-derived FSTL1, which induces cardiac angiogenesis through DIP2A-Smad2/3 signaling pathways[16].
Cardiac Protection Mechanisms
Follistatin-like 1 is highly expressed in cardiac tissue and released into serum following cardiac injury, where it functions as a cardiokine. The protein exerts multiple cardioprotective effects spanning from inhibiting inflammation through TGF-β modulation to preventing remodeling and fibrosis while promoting angiogenesis[17].
FSTL1 protects cardiomyocytes from ischemia/reperfusion injury through regulation of autophagy, with effects involving autophagy-associated protein modulation[18].
Inflammation and Immune Response Research
Pro-inflammatory Signaling
Follistatin-like protein 1 (FSTL1) influences inflammatory responses through multiple mechanisms. The protein improves NLRP3 inflammasome-mediated IL-1β secretion from monocytes and macrophages[19].
FSTL1 serum concentration increases substantially in bacterial sepsis and following lipopolysaccharide (LPS) administration. Within cells, FSTL1 localizes to mitochondria where it activates the mitochondrial electron transport chain and increases ATP production[19].
Anti-inflammatory Properties
In contrast to FSTL1’s pro-inflammatory actions, follistatin itself shows anti-inflammatory properties in various contexts. The protein alleviates synovitis and articular cartilage degeneration induced by inflammatory stimuli[20].
Follistatin substantially reduces macrophage infiltration into synovial membranes and inhibits proteoglycan erosion in articular cartilage. Treatment with activin-binding follistatin alters the profiles of key pro-inflammatory cytokines including TNF, IL-1β, and IL-6[20].
Exercise Physiology Research
Acute Exercise Responses
Circulating activins and follistatins increase approximately 10% to 21% following 45 minutes of various exercise types. Follistatin shows a 49% increase after anaerobic threshold exercise, with greater responses observed as exercise load increases[21].
The acute exercise-induced increase in follistatin appears more pronounced in sedentary individuals compared to lifelong exercisers. This differential response pattern provides insights for exercise adaptation research.
Training Adaptation Studies
Functional training influences irisin, follistatin, and myostatin levels in elderly individuals. Substantial increases in follistatin levels and reductions in myostatin levels occur following training interventions, accompanied by improvements in handgrip and shoulder girdle strength[22].
According to studies, the follistatin-to-myostatin ratio increases substantially in trained groups while decreasing in sedentary controls. This ratio shift represents a key marker in exercise adaptation research.
Intracellular Signaling Research
PI3K/Akt/mTOR Pathway
Follistatin promotes myoblast proliferation by activating the PI3K/Akt/mTOR signaling cascade. Treatment with pathway inhibitors abolishes follistatin-induced proliferative effects, confirming the requirement of this signaling network.
The protein induces expression of PI3K, Akt, mTOR, and downstream targets including 70-kDa ribosomal protein S6 kinase. In muscle tissue, follistatin-induced improvements in insulin action involve Akt signaling and PAK1 as follistatin targets[8].
Smad-Dependent Mechanisms
Activin controls follistatin transcription primarily through Smad3 action at intronic Smad-binding elements. FoxL2, a forkhead family member, acts as a modulator of Smad-mediated follistatin expression[23].
This represents an autocrine/paracrine feedback loop where activin induces its own antagonist through Smad-dependent mechanisms. Endothelial FSTL1 regulates pulmonary vascular development by modulating BMP/Smad signaling in lung tissue[24].
Potential In Vitro Research Applications
| Research Application | Primary Mechanism | Cell Models | Study Focus |
|---|---|---|---|
| Myoblast Proliferation | PI3K/Akt/mTOR pathway activation | Primary myoblasts, C2C12 cells | Muscle cell growth, fusion dynamics, satellite cell activation |
| Adipocyte Differentiation | Brown fat gene expression modulation | Brown preadipocytes, 3T3-L1 cells | Thermogenic protein expression, UCP1 induction, metabolic regulation |
| Osteogenic Differentiation | BMP antagonism, mesenchymal stem cell modulation | MSCs, bone marrow stromal cells | Bone formation pathways, mineralization, cell recruitment |
| Endothelial Cell Migration | Akt-eNOS signaling activation | HUVECs, endothelial progenitor cells | Angiogenesis, vascular repair, ischemic tissue models |
| Pancreatic Beta Cell Function | Activin neutralization, FSTL3 antagonism | Isolated islets, beta cell lines | Insulin secretion, glucose responsiveness, beta cell proliferation |
Sourcing High-Purity Research Follistatin
Quality is important when doing follistatin research. BioLongevity Labs offers research-grade Follistatin with independent verification from three certified laboratories.
Each batch is shipped with extensive documentation, including certificates of analysis (COAs), MSDS, and full analytical dossiers. All goods are manufactured in GMP facilities in the United States, with stringent quality control methods in place.
BioLongevity Labs’ research peptides consistently test greater than 99% purity. Every product is tested for purity using HPLC, molecular confirmation by LC-MS, and sterility and chemical contaminants.
All research peptides are solely for laboratory research purposes and are not intended for human consumption.
Scientific Reviewer
This research article has been scientifically reviewed and fact-checked by Dr. Ky H. Le, MD. Dr. Le earned his medical degree from St. George’s University School of Medicine and completed his residency training at Memorial Hermann Southwest Hospital. Board-certified in family medicine with experience in hospital medicine, he brings over two decades of clinical experience to reviewing research content and ensuring scientific accuracy.
About BioLongevity Labs
BioLongevity Labs supplies USA-made research peptides for in vitro laboratory applications. All compounds undergo independent third-party testing to verify purity and composition, with full certificates of analysis available for researchers requiring documentation. Browse our complete peptide catalog to find research-grade peptides for your laboratory needs.
References
- Lin SY, Craythorn RG, O’Connor AE, Matzuk MM, Girling JE, Morrison JR, et al. Female Infertility and Disrupted Angiogenesis Are Actions of Specific Follistatin Isoforms. The Endocrine Society; 2008. https://doi.org/10.1210/me.2006-0529
- Harrington AE, Morris-Triggs SA, Ruotolo BT, Robinson CV, Ohnuma S ichi, Hyvönen M. Structural basis for the inhibition of activin signalling by follistatin. Springer Science and Business Media LLC; 2006. https://doi.org/10.1038/sj.emboj.7601000
- Schneyer AL, Sidis Y, Gulati A, Sun JL, Keutmann H, Krasney PA. Differential Antagonism of Activin, Myostatin and Growth and Differentiation Factor 11 by Wild-Type and Mutant Follistatin. The Endocrine Society; 2008. https://doi.org/10.1210/en.2008-0259
- Hill JJ, Davies MV, Pearson AA, Wang JH, Hewick RM, Wolfman NM, et al. The Myostatin Propeptide and the Follistatin-related Gene Are Inhibitory Binding Proteins of Myostatin in Normal Serum. Elsevier BV; 2002. https://doi.org/10.1074/jbc.m206379200
- Pisconti A, Brunelli S, Di Padova M, De Palma C, Deponti D, Baesso S, et al. Follistatin induction by nitric oxide through cyclic GMP: a tightly regulated signaling pathway that controls myoblast fusion. Rockefeller University Press; 2013. https://doi.org/10.1083/jcb.2005070832003r
- Sun Y, Ge Y, Drnevich J, Zhao Y, Band M, Chen J. Mammalian target of rapamycin regulates miRNA-1 and follistatin in skeletal myogenesis. Rockefeller University Press; 2010. https://doi.org/10.1083/jcb.200912093
- Zhu J, Li Y, Lu A, Gharaibeh B, Ma J, Kobayashi T, et al. Follistatin Improves Skeletal Muscle Healing after Injury and Disease through an Interaction with Muscle Regeneration, Angiogenesis, and Fibrosis. Elsevier BV; 2011. https://doi.org/10.1016/j.ajpath.2011.04.008
- Li X, Liu H, Wang H, Sun L, Ding F, Sun W, et al. Follistatin could promote the proliferation of duck primary myoblasts by activating PI3K/Akt/mTOR signalling. Portland Press Ltd.; 2014. https://doi.org/10.1042/bsr20140085
- Braga M, Reddy ST, Vergnes L, Pervin S, Grijalva V, Stout D, et al. Follistatin promotes adipocyte differentiation, browning, and energy metabolism. Elsevier BV; 2014. https://doi.org/10.1194/jlr.m039719
- Perakakis N, Kokkinos A, Peradze N, Tentolouris N, Ghaly W, Tsilingiris D, et al. Follistatins in glucose regulation in healthy and obese individuals. Wiley; 2018. https://doi.org/10.1111/dom.13572
- Brown ML, Bonomi L, Ungerleider N, Zina J, Kimura F, Mukherjee A, et al. Follistatin and Follistatin Like‐3 Differentially Regulate Adiposity and Glucose Homeostasis. Wiley; 2011. https://doi.org/10.1038/oby.2011.97
- Han X, Møller LLV, De Groote E, Bojsen‐Møller KN, Davey J, Henríquez‐Olguin C, et al. Mechanisms involved in follistatin‐induced hypertrophy and increased insulin action in skeletal muscle. Wiley; 2019. https://doi.org/10.1002/jcsm.12474
- Fahmy-Garcia S, Farrell E, Witte-Bouma J, Robbesom-van den Berge I, Suarez M, Mumcuoglu D, et al. Follistatin Effects in Migration, Vascularization, and Osteogenesis in vitro and Bone Repair in vivo. Frontiers Media SA; 2019. https://doi.org/10.3389/fbioe.2019.00038
- Iemura S ichiro, Yamamoto TS, Takagi C, Uchiyama H, Natsume T, Shimasaki S, et al. Direct binding of follistatin to a complex of bone-morphogenetic protein and its receptor inhibits ventral and epidermal cell fates in early Xenopus embryo. Proceedings of the National Academy of Sciences; 1998. https://doi.org/10.1073/pnas.95.16.9337
- Xi Y, Hao M, Liang Q, Li Y, Gong DW, Tian Z. Dynamic resistance exercise increases skeletal muscle-derived FSTL1 inducing cardiac angiogenesis via DIP2A–Smad2/3 in rats following myocardial infarction. Elsevier BV; 2021. https://doi.org/10.1016/j.jshs.2020.11.010
- Horak M, Fairweather D, Kokkonen P, Bednar D, Bienertova-Vasku J. Follistatin-like 1 and its paralogs in heart development and cardiovascular disease. Springer Science and Business Media LLC; 2022. https://doi.org/10.1007/s10741-022-10262-6
- Kimura F, Sidis Y, Bonomi L, Xia Y, Schneyer A. The Follistatin-288 Isoform Alone Is Sufficient for Survival But Not for Normal Fertility in Mice. The Endocrine Society; 2009. https://doi.org/10.1210/en.2009-1176
- Fullerton PT, Monsivais D, Kommagani R, Matzuk MM. Follistatin is critical for mouse uterine receptivity and decidualization. Proceedings of the National Academy of Sciences; 2017. https://doi.org/10.1073/pnas.1620903114
- Yamada J, Tsuji K, Miyatake K, Matsukura Y, Abula K, Inoue M, et al. Follistatin Alleviates Synovitis and Articular Cartilage Degeneration Induced by Carrageenan. Wiley; 2014. https://doi.org/10.1155/2014/959271
- Increased Expression of Follistatin in Breast Cancer Reduces Invasiveness and Clinically Correlates with Better Survival. International Institute of Anticancer Research; 2017. https://doi.org/10.21873/cgp.20035
- Hofmann M, Schober-Halper B, Oesen S, Franzke B, Tschan H, Bachl N, et al. Effects of elastic band resistance training and nutritional supplementation on muscle quality and circulating muscle growth and degradation factors of institutionalized elderly women: the Vienna Active Ageing Study (VAAS). Springer Science and Business Media LLC; 2016. https://doi.org/10.1007/s00421-016-3344-8
- Elliott BT, Herbert P, Sculthorpe N, Grace FM, Stratton D, Hayes LD. Lifelong exercise, but not short-term high-intensity interval training, increases GDF11, a marker of successful aging: a preliminary investigation. Wiley; 2017. https://doi.org/10.14814/phy2.13343
- Blount AL, Vaughan JM, Vale WW, Bilezikjian LM. A Smad-binding Element in Intron 1 Participates in Activin-dependent Regulation of the Follistatin Gene. Elsevier BV; 2008. https://doi.org/10.1074/jbc.m709502200
- Jones KL, Mansell A, Patella S, Scott BJ, Hedger MP, de Kretser DM, et al. Activin A is a critical component of the inflammatory response, and its binding protein, follistatin, reduces mortality in endotoxemia. Proceedings of the National Academy of Sciences; 2007. https://doi.org/10.1073/pnas.0705971104
