Scientifically reviewed by
Dr. Ky H. Le, MD

The information presented in this article is for educational and research purposes only, intended for laboratory professionals, researchers and collaborators. This content does not constitute medical or clinical advice.
Lyophilization stands as the gold standard for preserving research peptides. This freeze-drying process converts liquid peptide solutions into stable solid-state compounds that maintain molecular integrity for years.
Understanding how lyophilization works—and why it’s so effective—helps research labs make informed decisions about peptide procurement, storage protocols, and reconstitution methods.
Highlights
- Lyophilization removes >99% of water from peptide solutions, creating a stable solid state that prevents most degradation pathways
- Properly lyophilized peptides remain stable for 3-5 years at -20°C with <1% residual moisture
- Cryoprotectants like trehalose protect peptide structure during the freeze-drying process and improve long-term stability
- Storage temperature matters: -80°C provides better preservation than -20°C for reconstituted peptide solutions
What Is Lyophilization?
Lyophilization—also called freeze-drying—is a controlled dehydration process that removes water from peptide formulations while preserving molecular structure.
The process reduces water content to less than 1%, transforming liquid samples into stable solid-state products. This dramatic reduction in moisture creates an environment where most degradation mechanisms simply cannot occur1.
The Three-Phase Process
Lyophilization happens in three distinct stages:
Freezing Phase
The peptide solution is cooled below its eutectic point, converting water to ice crystals. During this phase, the solution reaches 18-20% water content with all solutes concentrated in an amorphous phase separate from ice1.
Primary Drying
Vacuum pressure drops, allowing ice to sublimate directly from solid to vapor. This phase removes the bulk of the water through a solid-to-vapor transition that bypasses the liquid state entirely.
Secondary Drying
The sample is heated under deep vacuum to remove residual bound water through desorption. Properly processed products contain approximately 1% residual moisture after this final step2.
Lyophilized vs. Non-Lyophilized Peptides
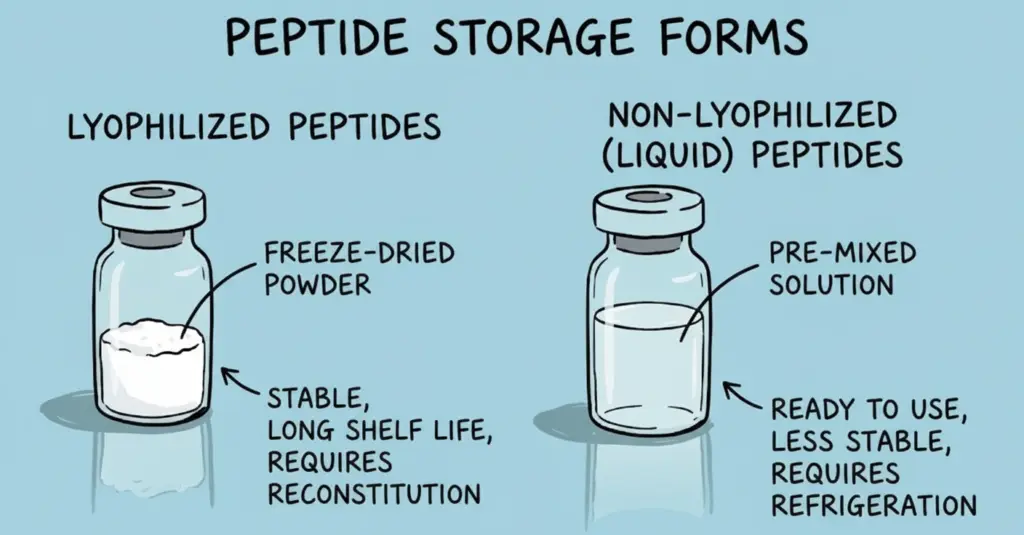
Peptides arrive in two primary forms: lyophilized powder or liquid solution. Each format serves different research needs.
Liquid Peptides
Non-lyophilized peptides come pre-dissolved and ready for immediate use. This convenience comes at a cost—liquid formulations typically require -80°C storage and have shorter shelf lives of weeks to months.
Peptides in solution face constant exposure to degradation mechanisms including oxidation, aggregation, and hydrolysis.
Lyophilized Peptides
Freeze-dried peptides offer superior stability with 3-5 year shelf life at -20°C. They require reconstitution before use but provide flexibility in choosing solvents and final concentrations.
The solid state protects against most degradation pathways that plague liquid formulations.
Laboratory Considerations
Most research labs prefer lyophilized peptides for inventory management and long-term stability. Liquid forms work best when immediate use is planned and ultra-cold storage is readily available.
Cost per experiment often favors lyophilized formats due to extended stability and reduced waste from expired stocks.
Why Lyophilization Protects Peptide Stability
Peptides face constant threats in aqueous solutions. Chemical bonds break down, side chains degrade, and aggregation occurs as molecules collide in solution.
Lyophilization stops these processes by removing the medium where degradation happens.
Prevention of Chemical Degradation
Most covalent degradation phenomena—including residue fragmentation and isomerization—require water. Remove the water, and these reactions cannot proceed1.
Oxidation represents another major degradation pathway that accelerates in aqueous environments. Peptides containing methionine, cysteine, or tryptophan residues are particularly vulnerable to oxidative damage when stored as solutions.
The solid state also dramatically reduces molecular mobility. Without water as a medium for movement, reactive groups cannot reach each other to form unwanted bonds or break existing ones.
Blocking Physical Degradation
Aggregation drops significantly because diffusion is minimized in solid versus liquid states. Peptide monomers cannot encounter each other when locked in a glassy matrix1.
This protection is critical because a peptide in solution faces constant molecular collisions that can trigger unwanted interactions. The liquid environment permits the diffusion necessary for aggregation to occur.
Research shows properly lyophilized peptide nanoparticles maintain high stability through temperature changes, freeze-thaw cycles, and extended storage3.
Creating a Protective Glass State
The lyophilization process creates an amorphous glassy state that locks peptides in place. This glass has extremely low molecular mobility, effectively freezing degradation processes in time4.
The glass transition temperature (Tg) of this matrix determines storage stability. Keep storage temperatures below Tg, and the protective glass remains intact.
Factors Affecting Lyophilized Peptide Stability
Multiple variables influence how well peptides survive the lyophilization process and subsequent storage.
Cryoprotectant Selection
Not all protective excipients perform equally.
Trehalose significantly outperforms sucrose, glucose, and dextran as a cryoprotectant. Studies found dextran provides almost no protective effect, while trehalose maintains peptide structure through both freezing and drying phases4.
The mechanism is straightforward. Cryoprotectants substitute for water molecules in maintaining protein structure, preventing collapse and denaturation as moisture disappears5.
Formulation Composition
Research demonstrates that trehalose-containing formulations preserved 85-86% of insulin structural integrity compared to formulations without protective excipients2.
Buffer choice matters too. pH shifts during freezing can denature peptides, so appropriate buffering agents must maintain pH throughout the process.
Process Parameters
Temperature control during each phase affects final product quality.
Annealing—holding samples at intermediate temperatures during freezing—reduced primary drying time by 38% while maintaining protein structure. This optimization speeds processing without compromising quality2.
Vacuum pressure, shelf temperature, and drying duration all require careful control. Too much heat denatures proteins. Insufficient drying leaves moisture that permits degradation.
Intrinsic Peptide Properties
Multiple factors affect peptide stability during lyophilization6:
- Amino acid sequence and composition
- Net charge and pH sensitivity
- Hydrophobicity and aggregation tendency
- Molecular weight and structural complexity
- Presence of disulfide bonds
- Surface-active regions
These intrinsic properties determine which protective strategies work best for specific peptide compounds.
Third-Party Tested, USA-Made, 99% Purity
Lyophilized peptides for laboratory applications. 99% purity, 100% USA-Made.
Storage Requirements for Lyophilized Peptides
Proper peptide storage and handling extends shelf life from months to years. Understanding how to store peptides correctly protects your research investment and ensures reproducible experimental results.
Temperature Guidelines
For Lyophilized Powder
Store at -20°C for optimal long-term stability. Research supports 3-5 year shelf life under these conditions with minimal degradation.
Some high-purity research peptides remain stable at 4°C for shorter periods, but -20°C provides the safest option for most compounds.
For Reconstituted Solutions
A longitudinal 10-month study concluded that -80°C provides optimal storage conditions for peptide solutions. The research recommended storing samples as separate aliquots to avoid repeated freeze-thaw cycles7.
Studies comparing -80°C to -150°C storage showed better recovery and viability at ultra-low temperatures, though brief temperature cycling between these ranges did not produce major changes8.
Storage Best Practices
| Storage Form | Temperature | Expected Stability | Notes |
|---|---|---|---|
| Lyophilized powder | -20°C | 3-5 years | Primary recommendation |
| Lyophilized powder | 4°C | 6-12 months | Short-term only |
| Reconstituted solution | -80°C | 1-3 months | Store as aliquots |
| Reconstituted solution | -20°C | 2-4 weeks | Avoid freeze-thaw |
| Reconstituted solution | 4°C | 1-7 days | Use quickly |
Container Selection
Use amber glass vials or high-quality polypropylene tubes. Protect from light and moisture with proper sealing.
Desiccants in storage containers help maintain low humidity, preventing moisture absorption that could trigger degradation.
Handling Precautions
Minimize exposure to room temperature. Brief temperature spikes during handling cause less damage than extended warm storage.
Allow vials to reach room temperature before opening to prevent condensation from forming on peptide powder.
Reconstitution Protocols
Reconstitution means dissolving lyophilized peptides into a solvent to restore them to their original active form for in vitro research applications9.
Solvent Selection
Sterile water works for most peptides. Some compounds require specific buffers or pH-adjusted solutions for optimal solubility.
Factors like pH, buffer composition, and temperature influence peptide solubility during reconstitution10. Check peptide specifications before selecting a reconstitution solvent.
Common reconstitution solvents include:
- Sterile water (most common)
- Phosphate buffered saline (PBS)
- Acetic acid solution (0.1-1%) for basic peptides
- Ammonium hydroxide for acidic peptides
- DMSO for highly hydrophobic compounds
Step-by-Step Reconstitution
1. Calculate Volume
Determine the volume needed to reach your target concentration. Most researchers prepare stock solutions at 1-10 mg/mL.
2. Prepare Solvent
Use fresh, sterile solvent at the appropriate temperature. Some peptides dissolve better at 37°C, while others prefer room temperature.
3. Add Solvent to Peptide
Never add peptide powder to solvent. Instead, slowly add solvent to the peptide vial to prevent foaming and aggregation.
4. Mix Gently
Swirl or rotate the vial gently. Avoid vortexing, which can denature sensitive peptides through shear stress and air interface exposure.
5. Verify Complete Dissolution
The solution should be clear with no visible particles. Cloudiness indicates incomplete dissolution or aggregation.
Reconstitution Troubleshooting
Slow Dissolution
Increase temperature slightly or add solvent in smaller increments with longer mixing periods between additions.
Cloudy Solution
This may indicate aggregation or precipitation. Try adjusting pH, changing solvents, or adding small amounts of detergent (if compatible with downstream applications).
Foaming Issues
Add solvent more slowly along the vial wall rather than directly onto the powder. Allow foam to settle before proceeding.
Quality Markers for Lyophilized Peptides
High-quality lyophilized research peptides meet specific criteria that indicate proper processing and storage.
Residual Moisture Content
Properly lyophilized products contain approximately 1% residual moisture2. Higher moisture levels permit degradation reactions that should not occur in the solid state.
Third-party testing through Karl Fischer titration provides accurate moisture measurement.
Structural Integrity
FTIR spectroscopy and circular dichroism confirm peptide structural maintenance after lyophilization2. These analytical methods detect secondary structure changes that indicate denaturation.
Mass spectrometry verifies molecular weight and identifies degradation products or modifications.
Purity Standards
Research-grade lyophilized peptides should meet >99% purity as verified by HPLC. Lower purity indicates incomplete synthesis, inadequate purification, or post-production degradation.
Certificate of Analysis (COA) documentation should include:
- HPLC chromatogram showing purity
- Mass spectrometry data confirming identity
- Residual moisture content
- Storage and handling recommendations
Research Applications
Lyophilized peptides serve diverse in vitro and ex vivo research applications across multiple fields.
Cell Culture Studies
Researchers use lyophilized peptides in cell signaling investigations, receptor binding assays, and pathway activation studies. The stable solid form permits precise dose preparation and reduces batch-to-batch variability.
Biochemical Assays
Enzyme-substrate interaction studies, inhibitor screening, and protein-protein interaction research all benefit from lyophilized peptide reagents. Long shelf life reduces the need for frequent reordering.
Drug Discovery Research
Pharmaceutical labs incorporate lyophilized peptides into high-throughput screening platforms. The format simplifies compound library management and automated liquid handling workflows.
Structural Biology
Crystallography and NMR studies often start with lyophilized peptide samples. The concentrated solid form facilitates preparation of the high-concentration solutions needed for structural analysis.
All BioLongevity Labs peptide products are for research use only and not intended for human or veterinary applications.
Sourcing High-Quality Lyophilized Peptides
Research outcomes depend on starting material quality. Several factors distinguish premium research peptides from lower-grade alternatives.
Manufacturing Standards
Look for USA GMP manufacturing facilities that follow Good Manufacturing Practices throughout production. Domestic manufacturing provides consistent quality control and comprehensive documentation.
Third-Party Verification
Triple third-party testing from independent certified laboratories confirms purity, identity, and quality. Each batch should include multiple analytical methods:
- HPLC for purity determination
- LC-MS for molecular weight confirmation
- Sterility and endotoxin testing
- Residual moisture analysis
Documentation Package
Complete analytical documentation should accompany every research peptide order. This includes batch-specific Certificates of Analysis showing exact purity, molecular weight, and storage recommendations.
Reconstitution Support
Suppliers should provide detailed reconstitution protocols specific to each peptide compound. Solvent recommendations, concentration guidelines, and stability data help researchers achieve reproducible results.
Scientific Reviewer
This research article has been scientifically reviewed and fact-checked by Dr. Ky H. Le, MD. Dr. Le earned his medical degree from St. George’s University School of Medicine and completed his residency training at Memorial Hermann Southwest Hospital. Board-certified in family medicine with experience in hospital medicine, he brings over two decades of clinical experience to reviewing research content and ensuring scientific accuracy.
About BioLongevity Labs
BioLongevity Labs manufactures research-grade peptides in USA GMP facilities with triple third-party testing verification. Every batch ships with comprehensive analytical documentation and detailed reconstitution protocols. Browse our lyophilized peptide vials to find compounds for your laboratory applications.
References
- Rathore N, Rajan RS. Current Perspectives on Stability of Protein Drug Products during Formulation, Fill and Finish Operations. Vol 24. Wiley; 2008:504-514. doi:10.1021/bp070462h
- Fonte P, Lino PR, Seabra V, Almeida AJ, Reis S, Sarmento B. Annealing as a Tool for the Optimization of Lyophilization and Ensuring of the Stability of Protein-Loaded PLGA Nanoparticles. Vol 503. Elsevier BV; 2016:163-173. doi:10.1016/j.ijpharm.2016.03.011
- Bruun TUJ, Andersson AMC, Draper SJ, Howarth M. Engineering a Rugged Nanoscaffold To Enhance Plug-and-Display Vaccination. Vol 12. American Chemical Society (ACS); 2018:8855-8866. doi:10.1021/acsnano.8b02805
- Bieganski RM, Fowler A, Morgan JR, Toner M. Stabilization of Active Recombinant Retroviruses in an Amorphous Dry State with Trehalose. Vol 14. Wiley; 1998:615-620. doi:10.1021/bp980057d
- Izutsu K ichi. Applications of Freezing and Freeze-Drying in Pharmaceutical Formulations. Springer Singapore; 2018:371-383. doi:10.1007/978-981-13-1244-1_20
- Zapadka KL, Becher FJ, Gomes dos Santos AL, Jackson SE. Factors Affecting the Physical Stability (Aggregation) of Peptide Therapeutics. Vol 7. The Royal Society; 2017:20170030. doi:10.1098/rsfs.2017.0030
- Planyavsky M, Huber ML, Staller NA, Müller AC, Bennett KL. A Longitudinal Proteomic Assessment of Peptide Degradation and Loss under Acidic Storage Conditions. Vol 473. Elsevier BV; 2015:11-13. doi:10.1016/j.ab.2014.11.020
- Yang J, Diaz N, Adelsberger J, et al. The Effects of Storage Temperature on PBMC Gene Expression. Vol 17. Springer Science and Business Media LLC; 2016. doi:10.1186/s12865-016-0144-1
- Cacho RA, Tang Y. Reconstitution of Fungal Nonribosomal Peptide Synthetases in Yeast and In Vitro. Springer New York; 2016:103-119. doi:10.1007/978-1-4939-3375-4_7
- Wearsch PA, Cresswell P. In Vitro Reconstitution of the MHC Class I Peptide-Loading Complex. Humana Press; 2012:67-79. doi:10.1007/978-1-62703-218-6_6
